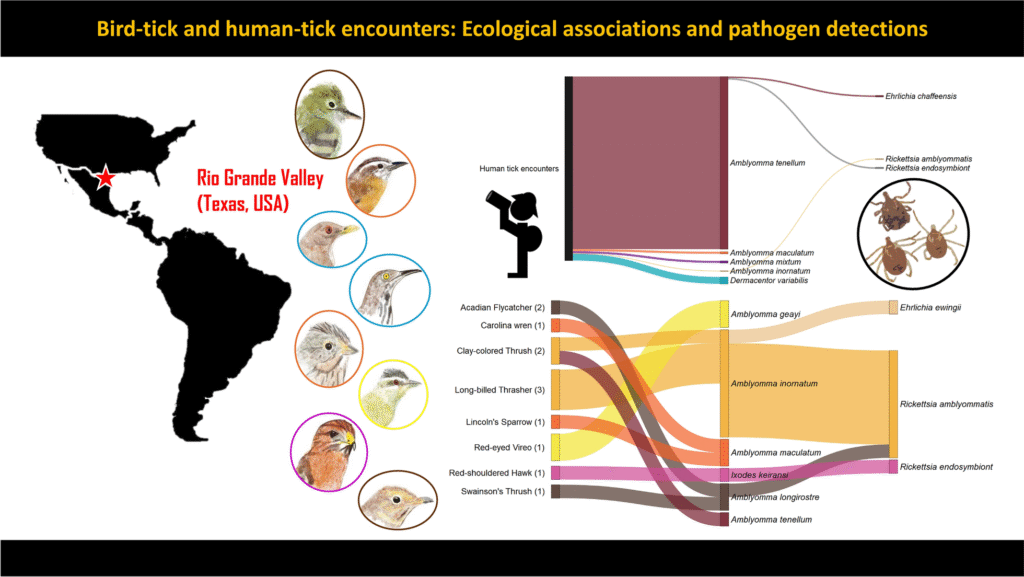
Rewrite a fully new long article using the information from
Marx GE, Spillane M, Beck A, Stein Z, Powell AK, Hinckley AF. Emergency department visits for tick bites — United States, January 2017–December 2019. Morb Mortal Wkly Rep. 2021;70:612–6.
Esteve-Gassent MD, Pérez de León AA, Romero-Salas D, Feria-Arroyo TP, Patino R, Castro-Arellano I, et al. Pathogenic landscape of transboundary zoonotic diseases in the Mexico–US border along the Rio Grande. Front Public Health. 2014;2:177.
Sonenshine DE. Range expansion of tick disease vectors in North America: implications for spread of tick-borne disease. Int J Environ Res Public Health. 2018;15:478.
Cohen EB, Auckland LD, Marra PP, Hamer SA. Avian migrants facilitate invasions of neotropical ticks and tick-borne pathogens into the United States. Appl Environ Microbiol. 2015;81:8366–78.
Karim S, Zenzal TJ, Beati L, Sen R, Adegoke A, Kumar D, et al. Ticks without borders: microbiome of immature neotropical tick species parasitizing migratory songbirds along northern Gulf of Mexico. Front Cell Infect Microbiol. 2024;14. https://doi.org/10.3389/fcimb.2024.1472598.
Shackelford CE, Rozenburg ER, Hunter WC, Lockwood MW. Migration and the migratory birds of Texas: who they are and where they are going -. 4th ed. Austin, Texas: Texas Parks and Wildlife; 2005.
Medlin JS, Cohen JI, Beck DL. Vector potential and population dynamics for Amblyomma inornatum. Ticks Tick-Borne Dis. 2015;6:463–72.
Williamson PC, Billingsley PM, Teltow GJ, Seals JP, Turnbough MA, Atkinson SF. Borrelia, Ehrlichia, and Rickettsia spp. in ticks removed from persons, Texas, USA. Emerg Infect Dis. 2010;16:441–6.
Maxwell SP, McNeely CL, Brooks C, Thomas K. Triangulating the new frontier of health geo-data: assessing tick-borne disease risk as an occupational hazard among vulnerable populations. Int J Environ Res Public Health. 2022;19:9449.
Thomas K, Brooks C, McNeely CL, Maxwell SP. Tick-Borne Disease risk and exposure among vulnerable populations in perceived non-endemic regions. Zoonotic Dis. 2022;2:111–6.
Texas DSHS. Ehrlichiosis [Internet]. Tex. Dep. State Health Serv. 2024 https://www.dshs.texas.gov/tick-borne-diseases/ehrlichiosis. Accessed 31 May 2024
Erickson TA, Mayes B, Murray KO, Gunter SM. The epidemiology of human ehrlichiosis in Texas, 2008–2017. Ticks Tick-Borne Dis. 2021;12:101788.
CDC. Ehrlichiosis epidemiology and statistics | CDC [Internet]. Cent. Dis. Control Prev. 2024 https://www.cdc.gov/ehrlichiosis/stats/index.html. Accessed 15 Nov 2021
Arroyave E, Quade B, Mendell NL, Blanton LS, Bouyer DH. Genetic characterization of a novel Ehrlichia chaffeensis genotype from an Amblyomma tenellum tick from South Texas, USA. Ticks Tick-Borne Dis. 2022;13:101990.
Erickson T, Gunter SM, Starke J, Murray KO. Evidence of locally acquired spotted fever group rickettsioses in Southeast Texas, 2008–2016. Zoonoses Public Health. 2018;65:897–901.
Mitchell EA, Williamson PC, Billingsley PM, Seals JP, Ferguson EE, Allen MS. Frequency and distribution of Rickettsiae, Borreliae, and Ehrlichiae detected in human-parasitizing ticks, Texas, USA. Emerg Infect Dis. 2016;22:312–5.
Tomassone L, Portillo A, Nováková M, de Sousa R, Oteo JA. Neglected aspects of tick-borne rickettsioses. Parasit Vectors. 2018;11:263.
Alfred JT, Mertins JW. New Identification Tools for the Nymphs of Three Amblyomma spp. Koch (Ixodida: Ixodidae). J Med Entomol. 2022;59:1607–14.
Cooley RA, Kohls GM. The Genus Amblyomma (Ixodidae) in the United States. J Parasitol. 1944;30:77–111.
Durden LA, Keirans JE. Nymphs of the genus Ixodes (Acari: Ixodidae) of the United States: taxonomy, identification key, distribution, hosts, and medical/veterinary importance. Maryland: Entomological Society of America; 1996.
Eads RB, Borom M. Host and distributional records for the tick Amblyomma Inornatum (Banks) (Acarina: Ixodidae), with descriptions of the immature stages. J Med Entomol. 1975;12:493–6.
Guzmán-Cornejo C, Robbins RG, Guglielmone AA, Montiel-Parra G, Pérez TM. The Amblyomma (Acari: Ixodida: Ixodidae) of Mexico: identification keys. Distribution and Hosts Zootaxa. 2011;2998:16–38.
Keirans JE, Durden LA. Illustrated key to nymphs of the tick genus Amblyomma (Acari: Ixodidae) found in the United States. J Med Entomol. 1998;35:489–95.
Kohls GM. Amblyomma imitator, a new species of tick from Texas and Mexico, and remarks on the synonymy of A. cajennense (Fabricius) (Acarina-Ixodidae). J Parasitol. 1958;44:430.
Nava S, Beati L, Dunlop J, Guglielmone AA. Reestablishment of Amblyomma tenellum Koch, 1844 (Acari: Ixodidae). Ticks Tick-Borne Dis. 2014;5:620–3.
Yunker CE, Keirans JE, Clifford CM, Easton ER. Dermacentor ticks (Acari: Ixodoidea: Ixodidae) of the New World: a scanning electron microscope atlas. Proc Entomol Soc Wash USA. 1986;88:609–27.
Beati L, Keirans JE. Analysis of the systematic relationships among ticks of the genera Rhipicephalus and Boophilus (Acari: Ixodidae) based on mitochondrial 12s Ribosomal DNA gene sequences and morphological characters. J Parasitol. 2001;87:32–48.
Murphy GL, Ewing SA, Whitworth LC, Fox JC, Kocan AA. A molecular and serologic survey of Ehrlichia canis, E. chaffeensis, and E. ewingii in dogs and ticks from Oklahoma. Vet Parasitol. 1998;79:325–39.
Jiang J, Stromdahl EY, Richards AL. Detection of Rickettsia parkeri and Candidatus Rickettsia andeanae in Amblyomma maculatum gulf coast ticks collected from humans in the United States. Vector-Borne Zoonotic Dis. 2012;12:175–82.
Salomon J, Fernandez Santos NA, Zecca IB, Estrada-Franco JG, Davila E, Hamer GL, et al. Brown Dog Tick (Rhipicephalus sanguineus Sensu Lato) infection with endosymbiont and human pathogenic rickettsia spp., in Northeastern México. Int J Environ Res Public Health. 2022;19:6249.
Fournier P-E, Dumler JS, Greub G, Zhang J, Wu Y, Raoult D. Gene sequence-based criteria for identification of new rickettsia isolates and description of rickettsia heilongjiangensis sp. nov. J Clin Microbiol. 2003;41:5456–65.
Margolis L, Esch GW, Holmes JC, Kuris AM, Schad GA. The use of ecological terms in parasitology (report of an ad hoc committee of the American Society of Parasitologists). J Parasitol. 1982;68:131–3.
Hook SA, Nawrocki CC, Meek JI, Feldman KA, White JL, Connally NP, et al. Human-tick encounters as a measure of tickborne disease risk in Lyme disease endemic areas. Zoonoses Public Health. 2021;68:384–92.
Maxwell SP, McNeely CL, Thomas K, Brooks C. Tick-borne surveillance patterns in perceived non-endemic geographic areas: human tick encounters and disease outcomes. Healthcare. 2021;9:771.
Mendell NL, Reynolds ES, Blanton LS, Hermance ME, Londoño AF, Hart CE, et al. Detection of Rickettsiae, Borreliae, and Ehrlichiae in ticks collected from walker county, Texas, 2017–2018. Insects. 2019;10:315.
Karpathy SE, Slater KS, Goldsmith CS, Nicholson WL, Paddock CD. Rickettsia amblyommatis sp. nov., a spotted fever group Rickettsia associated with multiple species of Amblyomma ticks in North, Central and South America. Int J Syst Evol Microbiol. 2016;66:5236–43.
Muñoz-Leal S, Marcili A, Fuentes-Castillo D, Ayala M, Labruna MB. A relapsing fever Borrelia and spotted fever Rickettsia in ticks from an Andean valley, central Chile. Exp Appl Acarol. 2019;78:403–20.
Romero LE, Binder LC, Marcili A, Labruna MB. Ticks and tick-borne rickettsiae from dogs in El Salvador, with report of the human pathogen Rickettsia parkeri. Ticks Tick-Borne Dis. 2023;14:102206.
Dolz G, Castro R, Jiménez-Rocha AE, Retamosa M, Alberti A. Strain diversity of Rickettsia amblyommatis in ticks infesting birds in the North Huetar conservation area of Costa Rica. Ticks Tick-Borne Dis. 2019;10:1109–12.
Bermúdez CSE, Félix ML, Domínguez AL, Kadoch N, Muñoz-Leal S, Venzal JM. Molecular screening for tick-borne bacteria and hematozoa in Ixodes cf. boliviensis and Ixodes tapirus (Ixodida: Ixodidae) from western highlands of Panama. Curr Res Parasitol Vector-Borne Dis. 2021;1:100034.
Olafson PU, Buckmeier BG, May MA, Thomas DB. Molecular screening for rickettsial bacteria and piroplasms in ixodid ticks surveyed from white-tailed deer (Odocoileus virginianus) and nilgai antelope (Boselaphus tragocamelus) in southern Texas. Int J Parasitol Parasites Wildl. 2020;13:252–60.
Sebastian PS, Flores FS, Saracho-Bottero MN, Tarragona EL, Venzal JM, Nava S. Molecular detection of rickettsial bacteria in ticks of the genus Ixodes from the Southern Cone of America. Acta Trop. 2020;210:105588.
Beati L, Nava S, Burkman EJ, Barros-Battesti DM, Labruna MB, Guglielmone AA, et al. Amblyomma cajennense (Fabricius, 1787) (Acari: Ixodidae), the Cayenne tick: phylogeography and evidence for allopatric speciation. BMC Evol Biol. 2013;13:267.
Nava S, Beati L, Labruna MB, Cáceres AG, Mangold AJ, Guglielmone AA. Reassessment of the taxonomic status of Amblyomma cajennense (Fabricius, 1787) with the description of three new species, Amblyomma tonelliae n. sp., Amblyomma interandinum n. sp. and Amblyomma patinoi n. sp., and reinstatement of Amblyomma mixtum Koch, 1844, and Amblyomma sculptum Berlese, 1888 (Ixodida: Ixodidae). Ticks Tick-Borne Dis. 2014;5:252–76.
Guglielmone AA, Petney TN, Robbins RG. Ixodidae (Acari: Ixodoidea): descriptions and redescriptions of all known species from 1758 to December 31, 2019. Zootaxa. 2020;4871:1–322.
Oliveira KA, Pinter A, Medina-Sanchez A, Boppana VD, Wikel SK, Saito TB, et al. Amblyomma imitator ticks as vectors of Rickettsia rickettsii. Mexico Emerg Infect Dis. 2010;16:1282–4.
Lockhart JM, Davidson WR, Stallknecht DE, Dawson JE, Howerth EW. Isolation of Ehrlichia chaffeensis from wild white-tailed deer (Odocoileus virginianus) confirms their role as natural reservoir hosts. J Clin Microbiol. 1997;35:1681–6.
CAPC. Parasite Prevalence Maps. Ehrlichiosis. Companion Anim. Parasite Counc. 2024 https://capcvet.org/maps. Accessed 5 Jun 2024.
Beall MJ, Alleman AR, Breitschwerdt EB, Cohn LA, Couto CG, Dryden MW, et al. Seroprevalence of Ehrlichia canis, Ehrlichia chaffeensis and Ehrlichia ewingii in dogs in North America. Parasit Vectors. 2012;5:29.
Gettings JR, Self SCW, McMahan CS, Brown DA, Nordone SK, Yabsley MJ. Local and regional temporal trends (2013–2019) of canine Ehrlichia spp. seroprevalence in the USA. Parasit Vectors. 2020;13:153.
Fudge JM, Boyanowski B, Page B, Liu S, Rogovskyy AS. Serological prevalence of six vector-borne pathogens in dogs presented for elective ovariohysterectomy or castration in the South central region of Texas. BMC Vet Res. 2020;16:381.
Gold BD. The role of birds as hosts for ticks, vectors of Borrelia burgdorferi, in eastern Texas [Internet] Dissertation. Texas State University; 2017. https://digital.library.txst.edu/server/api/core/bitstreams/33c050d5-8bc4-41c4-96ec-70e5f8510597/content
Roselli MA, Noden BH, Loss SR. Tick infestation of birds across a gradient of urbanization intensity in the United States Great Plains. Urban Ecosyst. 2022;25:379–91.
Tweit RC. Long-billed Thrasher (Toxostoma longirostre). In: Billerman SM, Keeney BK, Rodewald PG, Schulenberg TS, editors. Birds World. USA: Cornell Lab of Ornithology; 2020. https://doi.org/10.2173/bow.lobthr.01.
Hodo CL, Forgacs D, Auckland LD, Bass K, Lindsay C, Bingaman M, et al. Presence of diverse Rickettsia spp. and absence of Borrelia burgdorferi sensu lato in ticks in an East Texas forest with reduced tick density associated with controlled burns. Ticks Tick-Borne Dis. 2020;11:101310.
Estrada-Peña A, Binder LC, Nava S, Szabó MPJ, Labruna MB. Exploring the ecological and evolutionary relationships between Rickettsia and hard ticks in the Neotropical region. Ticks Tick-Borne Dis. 2021;12:101754.
Mukherjee N, Beati L, Sellers M, Burton L, Adamson S, Robbins RG, et al. Importation of exotic ticks and tick-borne spotted fever group rickettsiae into the United States by migrating songbirds. Ticks Tick-Borne Dis. 2014;5:127–34.
Fecchio A, Martins TF, Dias RI, Bell JA, Pinho JB, Silva VLDB, et al. Immature hard ticks infected with Rickettsia amblyommatis on breeding birds from Pantanal. Ticks Tick-Borne Dis. 2023;14:102121.
Richardson EA, Roe RM, Apperson CS, Ponnusamy L. Rickettsia amblyommatis in ticks: a review of distribution, pathogenicity, and diversity. Microorganisms. 2023;11:493.
Wright CL, Sonenshine DE, Gaff HD, Hynes WL. Rickettsia parkeri transmission to Amblyomma americanum by cofeeding with Amblyomma maculatum (Acari: Ixodidae) and potential for spillover. J Med Entomol. 2015;52:1090–5.
Dahlgren FS, Paddock CD, Springer YP, Eisen RJ, Behravesh CB. Expanding range of Amblyomma americanum and simultaneous changes in the epidemiology of spotted fever group rickettsiosis in the United States. Am J Trop Med Hyg. 2016;94:35–42.
Levin ML, Schumacher LBM, Snellgrove A. Effects of Rickettsia amblyommatis infection on the vector competence of Amblyomma americanum ticks for Rickettsia rickettsii. Vector Borne Zoonotic Dis Larchmt N. 2018;18:579–87.
Rivas JJ, Moreira-Soto A, Alvarado G, Taylor L, Calderón-Arguedas O, Hun L, et al. Pathogenic potential of a Costa Rican strain of ‘Candidatus Rickettsia amblyommii’ in guinea pigs (Cavia porcellus) and protective immunity against Rickettsia rickettsii. Ticks Tick-Borne Dis. 2015;6:805–11.
Snellgrove AN, Krapiunaya I, Scott P, Levin ML. Assessment of the pathogenicity of Rickettsia amblyommatis, Rickettsia bellii, and Rickettsia montanensis in a guinea pig model. Vector-Borne Zoonotic Dis. 2021;21:232–41.
Yen W-Y, Stern K, Mishra S, Helminiak L, Sanchez-Vicente S, Kim HK. Virulence potential of Rickettsia amblyommatis for spotted fever pathogenesis in mice. Pathog Dis. 2021;79:24.
Bonnet SI, Binetruy F, Hernández-Jarguín AM, Duron O. The tick microbiome: why non-pathogenic microorganisms matter in tick biology and pathogen transmission. Front Cell Infect Microbiol. 2017;7:236.
Nava S, Beati L, Venzal JM, Durden LA, Bermudez SE, Tarragona EL, et al. Description of two new species in the Ixodes ricinus complex from the new world (Acari: Ixodidae), and redescription of Ixodes affinis Neumann, 1899. Zootaxa. 2023;5361:53–73.
Oliver JH, Keirans JE, Lavender DR, Hutcheson HJ. Ixodes affinis neumann (acari: ixodidae): new host and distribution records, description of immatures, seasonal activities in georgia, and laboratory rearing. J Parasitol. 1987;73:646–52.
Narvaez ZE, Egizi AM, Price DC. First record of Ixodes keiransi (Acari: Ixodidae). J Med Entomol. 2024;61:798–801.
Zahler M, Gothe R, Rinder H. Genetic evidence against a morphologically suggestive conspecificity of Dermacentor reticulatus and D marginatus (Acari: Ixodidae). Int J Parasitol. 1995;25:1413–9.
Folmer O, Black M, Hoeh W, Lutz R, Vrijenhoek R. DNA primers for amplification of mitochondrial cytochrome c oxidase subunit I from diverse metazoan invertebrates. Mol Mar Biol Biotechnol. 1994;3:294–9.
Black WC, Piesman J. Phylogeny of hard- and soft-tick taxa (Acari: Ixodida) based on mitochondrial 16S rDNA sequences. Proc Natl Acad Sci. 1994;91:10034–8.
Haggerty TM, Morton ES. Carolina Wren (Thryothorus ludovicianus). In: Poole AF, editor. Birds World. NY, USA: Cornell Lab of Ornithology; 2020.
Panwar P, Deshwal A, Kannan R, Collar N, Spencer AJ. Clay-colored Thrush (Turdus grayi). In: Schulenberg TS, Keeney BK, Billerman SM, editors. Birds World. NY, USA: Cornell Lab of Ornithology; 2023.
Cornell Lab of Ornithology. All About Birds website. [Internet]. Birds. 2019. Available from: https://www.allaboutbirds.org. Accessed 7 Jun 2024
Dykstra CR, Morton ES, Hays JL, Crocoll ST. Red-shouldered Hawk (Buteo lineatus). In: Poole AF, editor. Birds World. NY, USA: Cornell Lab of Ornithology; 2020.
make sure the article is SEO-optimized following all the SEO Guides, from Focus Keyword to H2 and H3 titles and so on.
the article must explain all the details in a very clear and engaging structure.
very important: Use external links for keywords and sentences inside the article you will generate to boost the SEO.
Avoid writing any author related or donation related texts.
Avoid writing what you did in the articles because the article is published for visitors to read.